Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Kikuchi, Shin; Sakamoto, Kan*; Takai, Toshihide; Yamano, Hidemasa
Nihon Kikai Gakkai 2020-Nendo Nenji Taikai Koen Rombunshu (Internet), 4 Pages, 2020/09
In a postulated severe accidental condition of sodium-cooled fast reactor (SFR), eutectic melting between boron carbide (B C) as control rod element and stainless steel (SS) as control rod cladding or related structure may occur. Thus, behavior of B
C) as control rod element and stainless steel (SS) as control rod cladding or related structure may occur. Thus, behavior of B C-SS eutectic melting is one of the phenomena to evaluate the core disruptive accidents in SFR. In order to clarify the kinetic feature of B
C-SS eutectic melting is one of the phenomena to evaluate the core disruptive accidents in SFR. In order to clarify the kinetic feature of B C-SS eutectic melting process in the interface, the thinning test for SS crucibles using the pellets of B
C-SS eutectic melting process in the interface, the thinning test for SS crucibles using the pellets of B C or SS with low B
C or SS with low B C concentration were performed to obtain the rate constant with dependence of B
C concentration were performed to obtain the rate constant with dependence of B C concentration against SS. It was found that the rate constants of eutectic melting between SS and SS low B
C concentration against SS. It was found that the rate constants of eutectic melting between SS and SS low B C concentration were smaller than that of B
C concentration were smaller than that of B C-SS in the high temperature range. Besides, the rate constant of eutectic melting between SS and B
C-SS in the high temperature range. Besides, the rate constant of eutectic melting between SS and B C containing SS became small when decreasing the B
C containing SS became small when decreasing the B C concentration against SS.
C concentration against SS.
Nagaishi, Ryuji; Inoue, Masao; Hino, Ryutaro; Ogawa, Toru
Proceedings of 2014 Nuclear Plant Chemistry Conference (NPC 2014) (USB Flash Drive), 9 Pages, 2014/10
Since seawater has been used as a coolant for reactors and spent fuel pools in broken reactor buildings at Fukushima Daiichi NPS accident, radioactive contaminated water emitted following the accident has contained salt content of seawater at high concentrations, different from that at TMI-2 accident. Radiolysis of seawater leading to hydrogen generation and corrosion has been simulated and reported by several groups. However, the proposed radiolysis models cannot be always applied to water radiolysis at the wide range of salt concentrations present in the NPS, mainly because primary yields of radiolysis products of water and radiation-induced reactions are dependent on the salt concentration. In this study, the radiolytic behavior in diluted and concentrated systems of seawater was considered on the basis of results in steady state and pulse radiolysis experiments, in which the above salt effects were demonstrated from the obtained results.
JNC TN8400 2000-012, 33 Pages, 2000/04
The redox condition of near-field is expected to affect the performance of engineered barrier system. Especially, the oxygen initially existing in the pore space of compacted bentonites strongly affects the redox condition of the near-field. For assessing the influence of the oxygen, the transport parameters of it in the compacted bentonite and consumption process should be known. Therefore, following researches were conducted. In order to understand the diffusion of dissolved oxygen (DO) in compacted bentonite and to predict the effect of DO, the effective diffusion coefficients of DO in compacted sodium bentonite were measured by electrochemistry. As the results, the following relationship between the dry density of compacted sodium bentonite and the effective diffusion coefficient of DO in compacted sodium bentonite was derived: De=1.53 0.13
0.13 10
10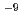 exp(-2.15
exp(-2.15 0.24
0.24 10
10 p) where De is the effective diffusion coefficient (m
p) where De is the effective diffusion coefficient (m s
s ) of DO in compacted sodium bentonite and
) of DO in compacted sodium bentonite and 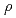 is the dry density (kg m
is the dry density (kg m ) of compacted sodium bentonite. The oxygen concentration in the bentonite is expected to be controlled by oxidation of pyrite as impurity in the bentonite. In order to investigate the above idea, the rates of pyrite oxidation by DO in compacted sodium bentonite were estimated from the experimental data on pyrite-bentonite systems usig the obtained effective diffusion coefficient of DO. The results show that the averages of the rate constants of pyrite oxidation by DO in the bentonite for dry densities of 0.8, 0.9, 1.0, 1.1 and 1.2
) of compacted sodium bentonite. The oxygen concentration in the bentonite is expected to be controlled by oxidation of pyrite as impurity in the bentonite. In order to investigate the above idea, the rates of pyrite oxidation by DO in compacted sodium bentonite were estimated from the experimental data on pyrite-bentonite systems usig the obtained effective diffusion coefficient of DO. The results show that the averages of the rate constants of pyrite oxidation by DO in the bentonite for dry densities of 0.8, 0.9, 1.0, 1.1 and 1.2 10
10 kgm
kgm were 1.38
were 1.38 0.32
0.32 10
10 , 1.10
, 1.10 0.24
0.24 10
10 , 1.16
, 1.16 0.35
0.35 10
10 , 9.36
, 9.36 2.23
2.23 10
10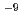 and 7.48
and 7.48 1.92
1.92 10
10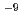 ms
ms , respectively. The relationship between the dry density (
, respectively. The relationship between the dry density (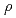 ) and the rate constant (k') was expressed as follows: k'=3.94
) and the rate constant (k') was expressed as follows: k'=3.94 1.06
1.06 10
10 exp(-1.33
exp(-1.33 0.28
0.28 10
10
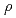 ) ...
) ...
 S and D
S and D S
SSato, K.*; *; *; *; *; Kurosaki, Yuzuru*; Takayanagi, Toshiyuki
Chemical Physics, 242(1), p.1 - 10, 1999/00
Times Cited Count:7 Percentile:22.56(Chemistry, Physical)no abstracts in English
 D,
D, P)+CH
P)+CH and CD
and CD reactions; The Role of nonadiabatic transitions on thermal rate constants
reactions; The Role of nonadiabatic transitions on thermal rate constantsTakayanagi, Toshiyuki; Kurosaki, Yuzuru*; Sato, K.*; *; *; *
Journal of Physical Chemistry A, 103(2), p.250 - 255, 1999/00
Times Cited Count:34 Percentile:71.42(Chemistry, Physical)no abstracts in English
 D,
D, P) with C
P) with C H
H and C
and C D
D between 225 and 292K
between 225 and 292KSato, K.*; *; *; *; *; Kurosaki, Yuzuru*; Takayanagi, Toshiyuki
Journal of Physical Chemistry A, 103(43), p.8650 - 8656, 1999/00
Times Cited Count:27 Percentile:64.06(Chemistry, Physical)no abstracts in English
 ZrO
ZrO packed bed
packed bedKawamura, Yoshinori; Enoeda, Mikio; Okuno, Kenji
Fusion Engineering and Design, 39-40, p.713 - 721, 1998/00
Times Cited Count:8 Percentile:57.28(Nuclear Science & Technology)no abstracts in English
 elimination from 2,3-dimethylbutane cation: (CH
elimination from 2,3-dimethylbutane cation: (CH )
) CHCH(CH
CHCH(CH )
)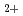
 (CH
(CH )
) C=C(CH
C=C(CH )
)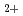 +H
+H ; An ab initio molecular orbital study
; An ab initio molecular orbital studyKurosaki, Yuzuru*; Takayanagi, Toshiyuki; Miyazaki, Tetsuro*
Journal of Molecular Structure; THEOCHEM, 452, p.209 - 218, 1998/00
no abstracts in English
 D,
D, P)+C
P)+C H
H and C
and C D
D reactions
reactionsTakayanagi, Toshiyuki; Kurosaki, Yuzuru*; *; *; *; Sato, K.*; *
Journal of Physical Chemistry A, 102(31), p.6251 - 6258, 1998/00
Times Cited Count:42 Percentile:78.25(Chemistry, Physical)no abstracts in English
 reaction
reactionTakayanagi, Toshiyuki; Kurosaki, Yuzuru*
Journal of Physical Chemistry A, 101(38), p.7098 - 7104, 1997/00
Times Cited Count:33 Percentile:73.31(Chemistry, Physical)no abstracts in English
*; *; Koike, Tadao; Miyata, Teijiro
JAERI-Tech 96-056, 59 Pages, 1996/12
no abstracts in English
Ogawa, Hiromichi; Takebe, Shinichi;
JAERI-Research 94-002, 12 Pages, 1994/07
no abstracts in English
Hashimoto, Kazuyuki; Kudo, Hiroshi; *; *
Radiochimica Acta, 63, p.167 - 171, 1993/00
no abstracts in English
*; Hirata, Masaru; Yahata, Taneaki
Journal of Nuclear Science and Technology, 28(8), p.739 - 747, 1991/08
no abstracts in English
JAERI-M 9336, 403 Pages, 1981/02
no abstracts in English


Journal of Nuclear Science and Technology, 16(3), p.221 - 222, 1979/00
Times Cited Count:1no abstracts in English
;
Bulletin of the Chemical Society of Japan, 50(8), p.2046 - 2050, 1977/08
Times Cited Count:8no abstracts in English
; Araki, Kunio
Journal of Polymer Science; Polymer Chemistry Edition, 14(11), p.2773 - 2783, 1976/11
no abstracts in English